Priority Areas - Analgesics: Opioid Burden
Opioid analgesics have long been used as the gold standard to treat severe pain, most notably for acute pain and in palliative care. The World Health Organisation analgesic ladder, which advocates a stepwise approach from non-opioid analgesics to mild opioids, followed by strong opioids, was developed with the aim of helping patients with cancer achieve freedom from pain.
Despite the analgesic ladder never having been validated for use in managing chronic, non-cancer pain, it has often been used as a guide to treatment in such scenarios resulting in patients receiving increasing doses of strong opioids. However such a simple approach is not appropriate for chronic pain which is highly complex.
With so many issues affecting the patient’s experience and reporting of chronic pain, it is not surprising that pain scores do not respond in any predictable fashion to opioids, and attempts to lower pain scores using opioids have led to overuse and adverse outcomes without any appreciable lowering of the chronic pain burden at the population level.
- The prescribing of opioids is often not the most appropriate or effective treatment option for patients with chronic pain, and can risk exposing patients to unnecessary harm.
- Despite the lack of evidence for use in chronic non-cancer pain, research has found an escalation of strong opioid prescribing in primary care, predominantly for non-cancer patients.
- In 2019, 121 deaths in Wales were registered as a result of drug-related poisoning, with opioids mentioned on the death certificate. Although this is a reduction from the 164 deaths registered in 2018, over 100 deaths annually have regularly been recorded in the last 10 years.
- Opioid analgesics have well established side effects including constipation, nausea and vomiting, and respiratory depression, and repeated administration may cause tolerance and dependence.
Trend in Opioid Burden
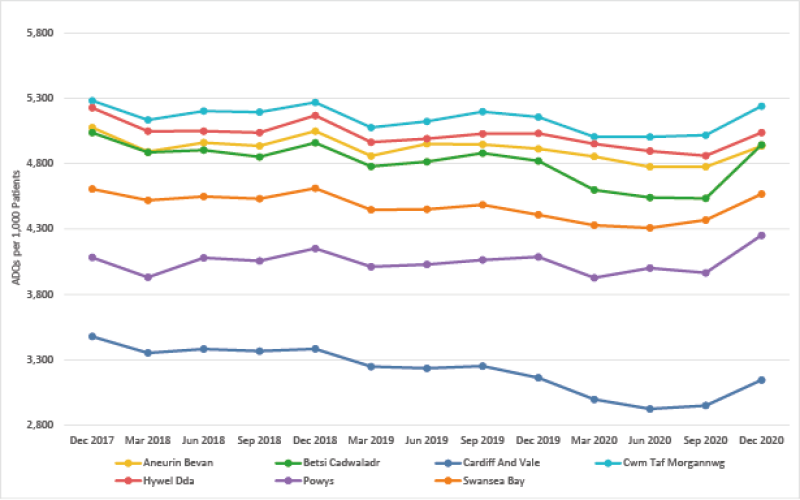
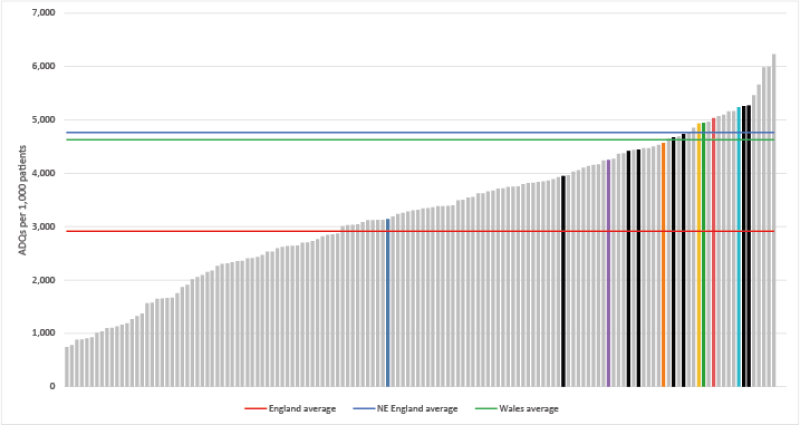
Opioid Burden ADQs per 1,000 patients - Quarter ending December 2020
Patients who do not achieve useful pain relief from opioids within two to four weeks are unlikely to gain benefit in the long term. A briefing paper by the BMA (Chronic Pain: supporting safer prescribing of analgesics), notes that too many people with chronic pain are prescribed opioids at high doses. The risk of harm increases substantially at high dose, and above an oral morphine equivalent daily dose of 120 mg, further benefit is unlikely. If benefit in pain reduction and improved function is not achieved at low dose, opioids should be discontinued, even if no other treatment is readily available. There is no evidence of efficacy of high dose opioids in long-term pain.
When making decisions about prescribing controlled drugs, consideration should be given to:
- the benefits of treatment;
- the risks of prescribing, including dependency, overdose and diversion;
- all prescribed and non-prescribed medicines taken by the patient, in addition to whether the person is opioid naïve.
A checklist from The Faculty of Pain Medicine is available to aid discussions with patients regarding opioid treatment.
How can changes be made?
- Ensure that when prescribing opioids, consideration is given to: the benefits of treatment; the risks of prescribing, including dependency, overdose and diversion; all prescribed and non-prescribed medicines the person is taking, and whether the person may be opioid naïve.
- If it is thought opioid therapy may play a role in a patient’s pain management, a trial should be initiated to establish whether the patient achieves a reduction in pain with the use of opioids – if not they should be stopped.
- Dose escalation should be limited as risk of harm rises as dose increases, especially if there is inadequate relief of pain. Above an oral morphine equivalent daily dose of 120 mg, further benefit is unlikely.
- Use The Royal College of Anaesthetists Faculty of Pain Medicine checklist to aid discussions regarding opioid treatment with patients.
- Conduct a search to identify patients on an oral morphine equivalent daily dose of ≥120 mg in order to undertake reviews.

