Case Study
Ann is 37 and attends her GP to talk about breast cancer risk. Ann’s sister who lives in Scotland has recently been diagnosed with breast cancer and is awaiting surgery. Ann is upset and is worried that she may be at risk. What do you do for Ann?

In the first instance you will need to deal with her emotions. You will need to obtain quite a lot of information from Ann in order to answer her questions and it is important this information is accurate.
If it is appropriate at this consultation you will need to take a full first and second-degree family history (including age at any diagnosis). If its not appropriate you may ask Ann to return, having spoken to her family members to gain the accurate information.
It would probably be appropriate to signpost the NICE patient information
Ann returns and has brought the information. She has 2 sisters (32 and 39) and a brother (42) her one sister is 39 at diagnosis her other two siblings are unaffected. Her mother was diagnosed with breast cancer at age 42 and died when she was 46, her father is still alive aged 73 with prostate cancer diagnosed aged 67. Her father has one sister who died, she had no children. Her mother was one of 4 sisters the surviving sisters are aged 65, 67 who are well with no significant PMH. A third maternal sister died aged 63 having been diagnosed with ovarian cancer at 61. Ann’s brother has two children, a boy aged 18 and a girl aged 14. Ann’s affected sister has two boys aged 11 and 12, her unaffected sister has a girl aged 8. Ann herself has three girls aged 11, 13 and 15. This generation of the family is unaffected. Can you draw the family tree and does Ann meet the criteria for referral to the Cancer Genetics Service for Wales?
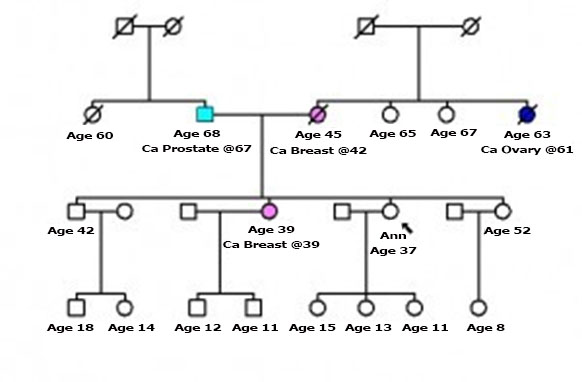
Ann is eligible for referral.
Ann attends the Cancer Genetics service and is given the following information regarding risk:- She has a 15% risk of being a carrier and she falls into the moderate risk category. She returns to her GP and wishes to take Tamoxifen to reduce her risk of developing breast cancer. She is concerned at the potential side effects. As her GP what would you discuss?

Ann will already have received counselling at the Cancer Genetics service. She will have a letter, which has been copied to you outlining the discussions. This letter would be a good stating point for the consultation, as the treatment options should be detailed there.
According to NICE guidance she is eligible for genetic testing, however in Wales this is not currently funded. Genetic testing of her affected sister would be more helpful for the whole family.
If Ann decides to take Tamoxifen you should discuss the fact that it is NICE endorsed but off licence. You may talk about the situation in the US where SERMs have been in regular use for this purpose. You should detail the risks, which would be an increased risk of endometrial cancer, DVT, PE and CVA. Cataract development risk is slightly increased also. Side effect profile should be discussed including menopausal symptoms. You may need to discuss contraception and smoking (as appropriate).
If the decision is made to prescribe signs and symptoms associated with the risks should be detailed. The treatment course is 5 years and this should be discussed with Ann and recorded to enable the prescription to be terminated at the appropriate time.
Are there any implications in your practice?
This will obviously depend on practice policy for prescribing off licence. This is covered by Good practice in prescribing and managing medicines and devices (2013) GMC.
It may be worthwhile bringing the information contained in this web resource to a practice meeting to raise awareness and discuss the implications.

