Safety - Yellow Cards
The Yellow Card Scheme is vital in helping the MHRA monitor the safety of all healthcare products in the UK to ensure they are acceptably safe for those that use them.
Yellow Card reporting supports the identification and collation of Adverse Drug Reactions (ADRs) which might not have been known about before.
ADRs are a significant clinical problem, increasing morbidity and mortality and studies have shown that ADRs are the cause of around 6.5% of hospital admissions in adults. An ADR has been defined as “a response to a medicinal product that is noxious and unintended, resulting not only from the authorised use of a medicinal product at normal doses but also from medication errors and use outside the terms of the marketing authorisation, including the misuse and abuse of the medicinal product”.
The aim of this NPI is to encourage an increase in reporting and ensure it is embedded as routine practice.
- ADRs are a significant clinical problem, increasing morbidity and mortality.
- The Yellow Card Scheme is vital in helping the MHRA monitor the safety of all healthcare products in the UK to ensure they are acceptably safe for those that use them.
- Yellow Card reporting supports the identification and collation of ADRs which might not have been known about before.
Trend in GP Yellow Card Reporting
Although the number of reports is generally increasing, there are concerns that it is not yet routine practice for all healthcare professionals.
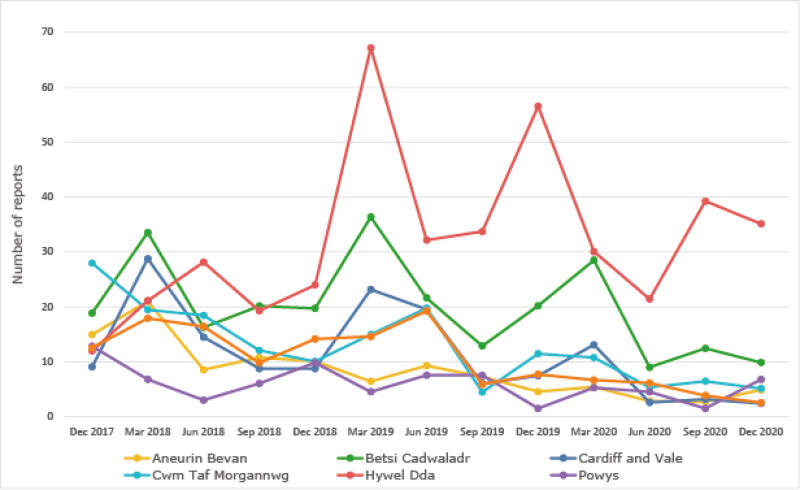
Please note, prior to the quarter ending June 2019, data displayed for Cwm Taf Morgannwg and Swansea Bay UHBs relate to the former health boards of Cwm Taf and Abertawe Bro Morgannwg, respectively.
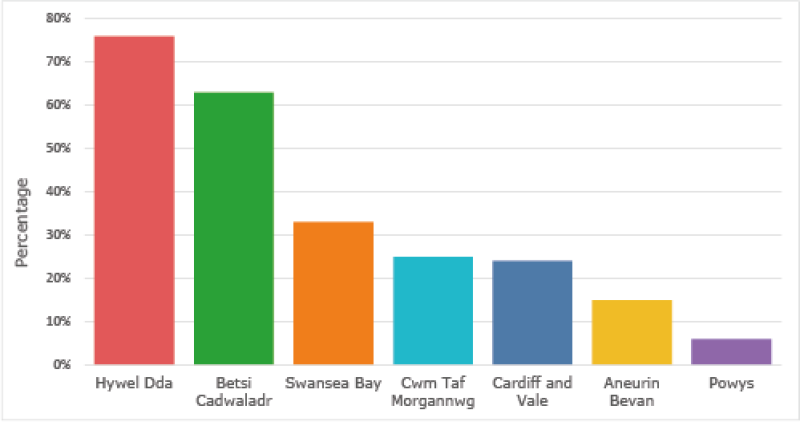
The primary care target for this indicator is for each GP practice to submit one or more Yellow Cards per 2,000 practice population annually.
The graph below shows the percentage of GP practices in each health board who met the target during 2019-2020.
Percentage of GP practices meeting the target of one Yellow Card per 2,000 practice population 2019–2020
The effectiveness of the Yellow Card scheme to detect new drug safety signals is dependent upon reporting of suspicions and observations.
Yellow Cards can be used for reporting suspected ADRs to medicines, vaccines and herbal products.
You should report all suspected ADRs that are:
- Serious, medically significant, result in harm, or are associated with medication errors where harm occurs.
- Serious events are those which are fatal, life-threatening, a congenital abnormality, disabling or incapacitating, or resulting in hospitalisation
In addition, you should report all suspected ADRS that are:
- Associated with newer drugs and vaccines, noted by the black triangle, irrespective of whether they are serious or not. The most up-to-date list of black triangle medicines is available on the MHRA website.
- If in doubt whether to report or not, please complete a Yellow Card. Patients can also be encouraged to complete Yellow Cards; the system can detect duplicate entries, so please complete a Yellow Card even if you think it has already been reported. Additional information can add further details to the case.
You only need to suspect that an ADR was caused by a medicine in order to report it.
How can changes be made?
- Yellow Card reports can be completed:
- Online: mhra.gov.uk/yellowcard
- Using the free Yellow Card app
- Through the INPS Vision prescribing system
- By writing to FREEPOST YELLOW CARD
- By emailing: yellowcard@mhra.gov.uk
- By downloading a from the website
- You only need to suspect that an adverse drug reaction was caused by a medicine to report it.
- Don’t assume someone else will report an adverse drug reaction you witness.
- Reports can be made for all medicines including vaccines, blood factors and immunoglobulins, herbal medicines and homeopathic remedies, and all medical devices available on the UK market.
- Further information can be gained from the Yellow Card Centre Wales.

