Priority Areas - Analgesics: Gabapentin and Pregabalin
Gabapentin and Pregabalin are licensed for peripheral neuropathic pain and epilepsy; pregabalin is also licensed for generalised anxiety disorder.
Prevalence data suggests that the majority of gabapentin and pregabalin prescribed is for neuropathic pain, and both are recommended by NICE as first-line options in the treatment of neuropathic pain. Other first line options include amitriptyline and duloxetine.
Cases of misuse, abuse and dependence have been reported with both gabapentin and pregabalin, and concerns over these issues have been raised by the Advisory Council on the Misuse of Drugs. This led to the scheduling of gabapentin and pregabalin under the Misuse of Drugs Act as Class C substances.
Caution should be exercised in prescribing either drug for patients with a history of substance abuse, and the patient should be monitored for symptoms of misuse or dependence. Pregabalin may have a higher abuse potential than gabapentin due to its rapid absorption, faster onset of action and higher potency.
- While there is a recognised place in neuropathic pain management for gabapentin and pregabalin, there are concerns regarding the risks associated with dependence, diversion and misuse.
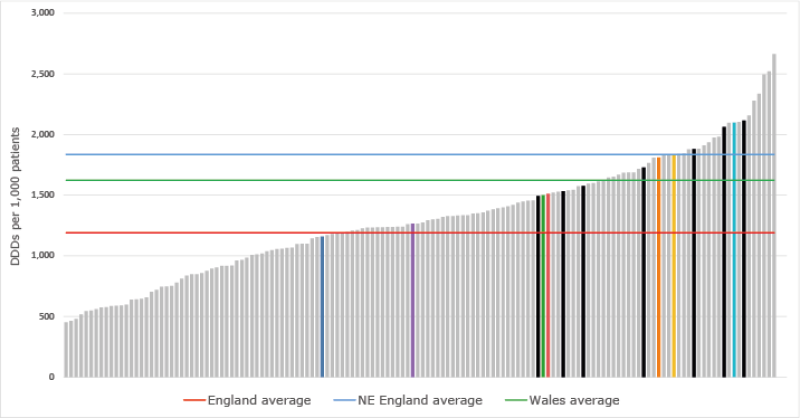
- There has been increasing use of gabapentin and pregabalin in primary care over the last 5 years across Wales. In addition, prescribing is high in comparison with England.
- There has been an increase in the number of deaths where gabapentin or pregabalin was mentioned on the death certificate in England and Wales, from 64 deaths registered in 2014 to 333 deaths registered in 2019.
Trend in Gabapentin and Pregabalin prescribing
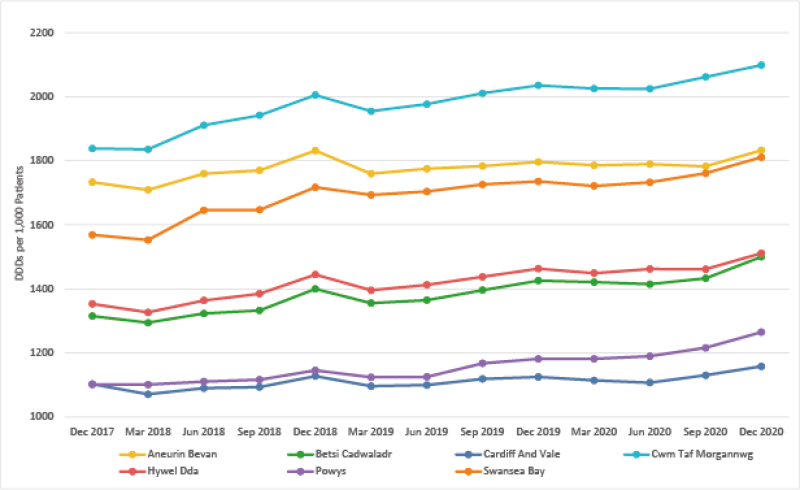
Gabapentin and Pregabalin DDDs per 1,000 patients – Quarter ending December 2020
Neuropathic pain management is often complex and prescribers need to make evidence based, informed decisions based on the individual needs of the patient. Prescribers should be aware not only of the potential benefits of gabapentin and pregabalin, but also that they may be misused or diverted. Individuals who are misusing analgesics need to be distinguished from those who are using higher or more frequent doses because their symptoms are not being adequately treated.
In order to assess whether the patient’s pain is neuropathic in nature, a number of pain scales are available, including the Leeds assessment of neuropathic symptoms and signs, and the Pain Detect pain questionnaire. Use of pain scales will also assist in determining response to treatment and should be used at each review.
How can changes be made?
- Use a pain scale (for example, the Leeds assessment of neuropathic symptoms and signs [LANSS]) to assess whether the patient’s pain is neuropathic in nature. This will also assist in determining response to treatment.
- Inform patients that response to drug treatment in neuropathic pain is often inadequate, with no more than 40–60% of people obtaining partial pain relief.
- Once treatment has commenced, NICE recommends early assessment followed by regular reviews to assess and monitor effectiveness including pain control, adverse effects and continued need.
- Reduce and stop the gabapentin or pregabalin if the patient has not shown sufficient benefit within eight weeks of reaching the maximum tolerated dose.
- Exercise caution in prescribing gabapentin or pregabalin for patients with a history of substance abuse.

